
The discovery of francium completed humankind’s discoveries of naturally occurring elements.Īll elements discovered since then have been discovered when the element has been produced in the laboratory.Ī nucleus emits a beta-particle (an electron) and an antineutrino. With 87 protons the new element belonged in Group 1 of the periodic table, joining the other five alkali metals: lithium, sodium, potassium, rubidium and cesium. The daughter nucleus formed was a previously undiscovered element which she chose to call francium to honor her home country, France. Perey had discovered that actinium-227 could decay by emitting a helium nucleus (also called an alpha particle) from its own nucleus. Perey discovered that about 1% of actiniums radioactivity was caused by it emitting alpha particles, not beta particles. She produced an ultra-pure actinium sample and studied its radiation. Perey was curious about this finding, and already being an expert on actinium related work, she decided to carry out her own experiments on actinium. 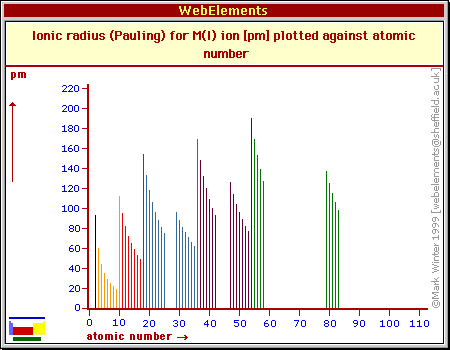
The discovery began, in 1935, when Perey aged 26, read a research paper claiming that American scientists had discovered beta particles being emitted by actinium which had a higher amount of energy than normal. The element takes its name from the country of its discovery – France. The discovery took place at the Curie Institute in Paris. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Francium was discovered by Marguerite Perey in 1939 when she was researching the radioactive decay of actinium-227. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). If you have any questions, then ask me in the comments below.Īlso let me know, have you downloaded the HD image or not? Free Gift for you: Interactive Periodic Table I hope this article has helped you in solving your problem. (Note: Above mentioned radii are the van der Waals radius.) 
The list of Atomic radius of elements in periodic table is mentioned below. Now let’s see the atomic radius chart for elements of Periodic table. 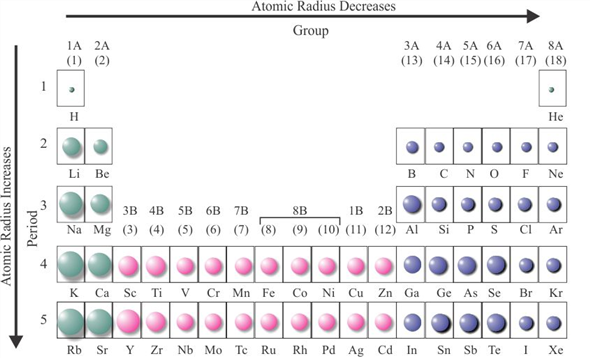
Must see: Atomic size trend in Periodic table (where you will come to know why and how the size of atoms changes across a period and along a group)
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
